|
By measuring the ratio of different isotopes present within a sample, scientists can calculate how long it has been since the material was last in equilibrium with its environment. One of the most common uses of isotopes is in radiometric dating, which is used to determine the age of rocks and fossils. Isotopes can also be used in a variety of applications, such as radiometric dating, medical imaging, and nuclear energy. By studying the properties of different isotopes, scientists can gain a better understanding of how atoms interact with each other and the environment around them. Isotopes play an important role in a variety of scientific fields, including chemistry, biology, and physics. For example, carbon has three different isotopes: carbon-12, carbon-13, and carbon-14, which all have the same number of protons (6) but different numbers of neutrons (6, 7, and 8, respectively). Neutrons also play a role in determining an atom’s isotope, which refers to a specific variation of an element that has a different number of neutrons than the standard version. This helps to keep the positively charged protons within the nucleus from repelling each other, which could cause the atom to become unstable and potentially decay. While protons and electrons both carry a charge, neutrons are neutral, meaning they do not have a charge. 
Neutrons are an essential component of the atomic structure, playing a crucial role in determining an atom’s stability and reactivity. The Significance of Neutrons in the Nucleus For example, if an atom has a mass number of 13 and an atomic number of 6, we can determine the number of neutrons in its nucleus by subtracting 6 from 13, giving us a total of 7 neutrons. 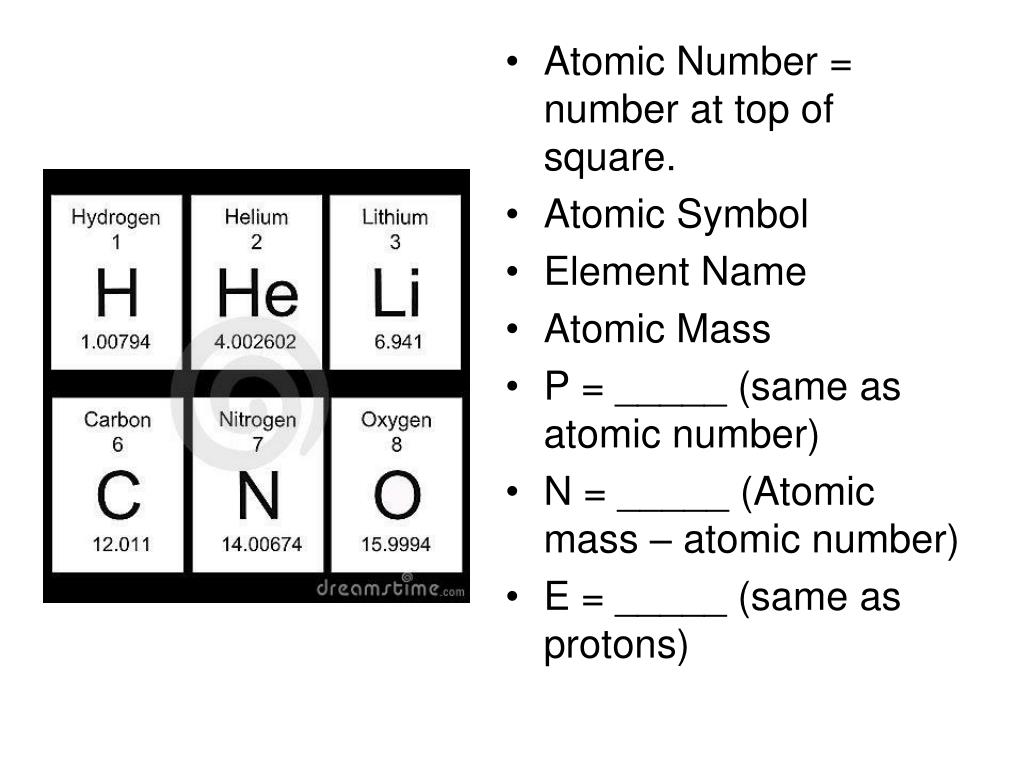
This means that the number of neutrons within an atom can be determined by subtracting the atomic number (which represents the number of protons) from the mass number. The mass number of an atom is the sum of the number of protons and neutrons within its nucleus. For example, an atom with six protons is always carbon, while an atom with eight protons is always oxygen. The number of protons found within an atom’s nucleus determines its atomic number, which can be used to identify the element. Protons and neutrons are found within the nucleus of an atom, while electrons move around the outside in shells or energy levels. The atomic structure of an atom is made up of three different particles: protons, neutrons, and electrons. Understanding the Basics of Atomic Structure 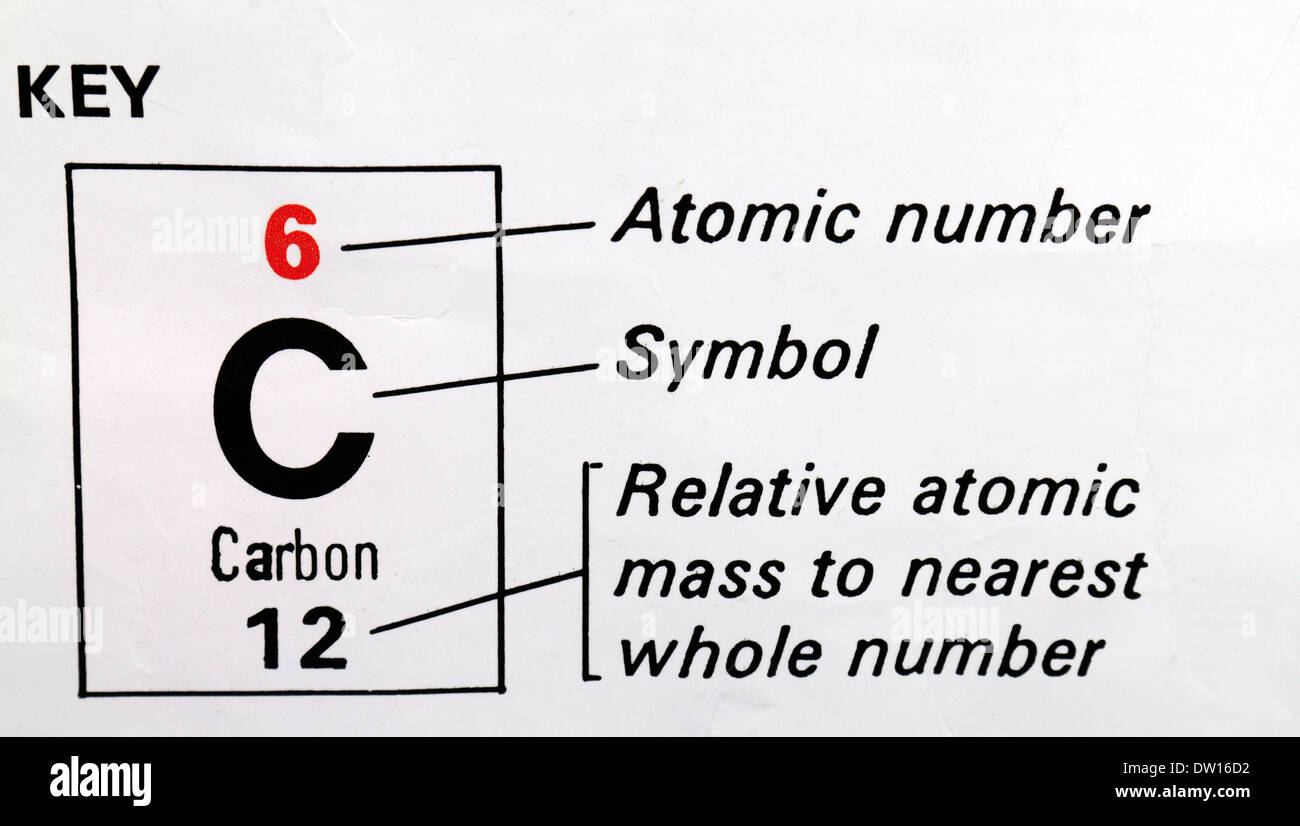
This article will examine an atom with a mass number of 13 and an atomic number of 6, and explore how many neutrons are in its nucleus. The atomic number of an atom refers to the number of protons found within its nucleus, while the mass number represents the total number of protons and neutrons present. When discussing atoms, one of the most important pieces of information to consider is the number of protons, neutrons, and electrons that make up its structure. An Atom’s Mass Number Is 13 And Its Atomic Number Is 6 – How Many Neutrons Are In Its Nucleus? There are 7 neutrons in the nucleus of an atom with a mass number of 13 and an atomic number of 6. The number of neutrons in the nucleus of an atom with a mass number of 13 and an atomic number of 6 is 7.ĥ. This atom has 7 neutrons in its nucleus.Ĥ. The number of neutrons in the nucleus of this atom is 7.ģ. An atom with a mass number of 13 and an atomic number of 6 has 7 neutrons in its nucleus.Ģ. 
Calculating Neutrons in an Atom: A Guide to Atomic Structure An atom’s mass number is 13 and its atomic number is 6.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
